|
217 Ft. Johnson Rd. |
|
Featured
Species! |
|||||||
| Paradella dianae - around the world in 20 years
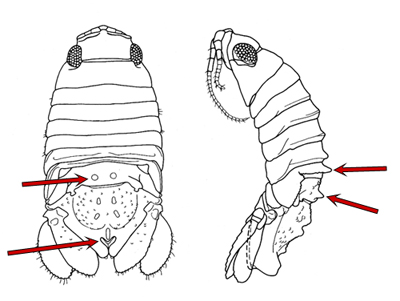
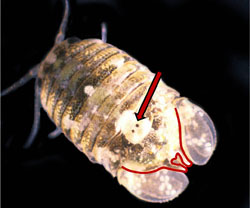
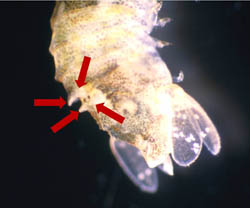
Paradella dianae is a species of crustacean that was accidentally introduced to the southeast coast of the U.S. in the early 1980s. It was first discovered by SCDNR divers who were studying the jetties that were being built at Murrells Inlet at that time. As they made repeated dives on the jetty stones below the low tide level, to carefully and systematically quantify the flora and fauna, divers noticed hundreds of small creatures clinging tightly to their neoprene wetsuits when they climbed from the water back onto the dive boat. It took a lot of effort to remove them, even under the heavy spray of freshwater from a garden hose back at the dock. It turns out that these pesky animals were isopods that are native to the Pacific coasts of North and Central America. They were probably carried to our coast on the outside surfaces of oceangoing ships, and they have hitchhiked around the world among the fouling growth that builds up over time on these ship�s hulls. Although they aren�t particularly conspicuous to the casual observer, isopods are an important part of many coastal communities, as this is especially true for those that live on hard surfaces that are continuously submerged in high salinity seawater for a reasonably long period of time (e.g. floating docks, pilings and jetties). You can learn more about this interesting group of crustaceans by going to the archived 'Featured Species' at http://www.dnr.sc.gov/marine/sertc/Isopod%20Crustaceans.pdf Description and Biology:Paradella dianae is a dorso-ventrally flattened, yellowish and brown colored sphaeromatid isopod. It somewhat resembles the terrestrial isopods that are commonly known as pill bugs or roly pollies. Mature males and females differ in shape and size. Adults are approximately 3 to 5 mm [~0.1 to 0.2 inches] in length, and males are larger than females (Glynn, 1970; Harrison and Holdich, 1982). Males can be easily distinguished from females by the presence of a heart-shaped indentation (foramen) at the posterior end of the animal�s midline and by the prominent tubercles on its back (Figs. 1-3).
Figure 1. Male Paradella dianae, dorsal and lateral views. Arrows point to features that distinguish this species from other local species in the family Sphaeromatidae (from Harrison & Holdich, 1982).
Figure 2. Photographs of male Paradella dianae from the dorsal and lateral aspects, with the distinguishing features shown in Fig. 1 indicated.
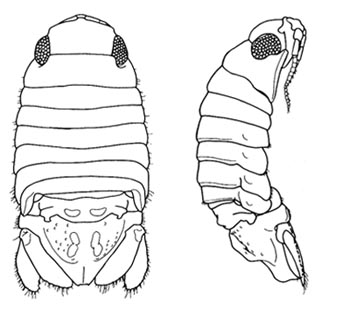
Figure 3. Female Paradella dianae, dorsal and lateral views. Note lack of heart-shaped foramen and dorsal tubercles seen in males, as shown in Figs. 1&2 (from Harrison & Holdich, 1982). Paradella dianae is commonly found among intertidal algae on a variety of substrates, including rock, mud and man-made surfaces like jetties, buoys and piers. The species is often rather secretive, hiding among the heavy growth of marine invertebrates, under stones, or in the shells of dead barnacles or polychaete tubes, especially when the tide has gone down. For food, it grazes on the algae and bacterial film that is attached to the surface (Harrison and Holdich, 1982). P. dianae is known to survive at temperatures as low as 14°C [57.2°F] (Nelson and Demetriades, 1992), although its established range on the US Atlantic coast suggests that it tolerates temperatures lower than that. It can survive in a wide range of salinities, from full-strength ocean water to estuarine water diluted to less than half of that (Fulani, 1996; Rodriguez et al., 1992), and it is also known to withstand heavy pollution (Pires, 1980). This species has a rather unusual life history, since it starts life as a female and changes some time later into a male. This unusual lifestyle, known to scientists as protogynous hermaphroditism, was discovered by Kensley and Schotte (1999). After a male and a female P. dianae copulate, eggs are deposited in a brood pouch on the female’s underside. The number of eggs may range from 14 to 25 per brood (García-Guerrero and Hendrickx, 2005). When the eggs have reached the appropriate stage of development, small juvenile isopods are released into the surrounding environment. In Florida populations, P. dianae has two peaks in abundance during the year; one occurs in sometime from late winter to early spring, and the second happens in late summer to early autumn (Nelson and Demetriades, 1992). However, in its native range, females bearing eggs (gravid) were only collected during the summer, with peaks off egg production in the month of June (García-Guerrero and Hendrickx, 2005). Similar species: Paradella dianae resembles several other species in the family Sphaeromatidae, which is represented in the SAB by the genera Ancinus, Cassidinidea, Paracerceis, Sphaeroma and Exosphaeroma. Kensley and Schotte (1989) provide keys to the four sphaeromatid subfamilies and to species in each of these genera.
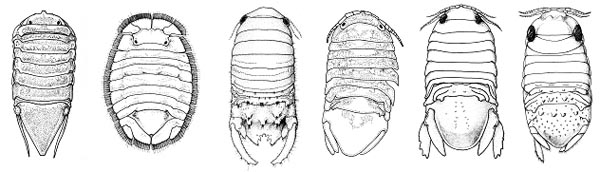
Figure 4. Male sphaeromatid isopods of South Carolina. A: Ancinus sp.; B: Cassidinidea ovalis;C: Paracerceis caudata; D: Exosphaeroma diminutum; E: Sphaeroma quadridentaum; F: Sphaeroma terebrans (also nonindigenous in South Carolina). Illustrations A-C, E, F are from Kensley & Schotte, 1989, Smithsonian Institution Press; D is from Menzies & Frankenberg, 1966, University of Georgia Press. Native range: Paradella dianae is a native of the Pacific coast of North America, from Ventura County, California to Michoacan, Mexico and along the west coast of the Gulf of California from Guaymas Sonora to Sayulita, Nayarit, Mexico (Espinosa-Pérez and Hendrickx, 2001; Iverson, 1974). Introduced range: A real world traveler, Paradella dianae has been found in many locations around the globe. Unable to spread so far under its own power, the isopod has most likely been accidently introduced to new locations by hitchhiking on the hulls and other exterior surfaces of ships. The species is now known from such far-flung locations as the Marshall Islands (Glynn, 1970), both coasts of Australia (Harrison and Holdich, 1982), the Atlantic coast of Spain (Rodriguez et al., 1992), the Mediterranean Sea (Atta, 1991; Forniz & Maggiore, 1985), the Arabian Sea (Javed and Ahmed, 1987) and Hong Kong (Bruce, 1990). In the Western Atlantic Ocean, P. dianae is known from Brazil (Pires, 1980), Venezuela (Glynn, 1970), Texas (Clark and Robertson, 1982), and Florida, including Key West (Kensley and Schotte, 1989). At the time of its earliest discovery in South Carolina on the jetty rocks at Murrells Inlet (reported by Van Dolah et al., 1984 as Paradella quadripunctata), it initially reached abundances as great as 1500 individuals in one square meter. Subsequent collections of Paradella dianae in South Carolina have been made at Charleston Harbor, Sullivans Island, North Inlet and Murrells Inlet (Fox and Ruppert, 1985; Knott, unpublished data). History of dispersal: The first record of Paradella dianae outside of its native range was from the Marshall Islands in 1967 (Glynn, 1970). Four years later the species was found in Townsville, Queensland, Australia, and then in 1980 Furlani (1996) observed it in Western Australia. On the other side of the Indian Ocean, P. dianae was found in the Arabian Sea in 1984 (Javed and Ahmed, 1987), then in the Mediterranean Sea (Forniz & Maggiore, 1985), Hong Kong in 1986 (Bruce, 1990) and along the Atlantic coast of Spain in 1988 (Rodriguez et al., 1992). The first report of P. dianae in the Atlantic Ocean was from Mayagüez Bay, Puerto Rico in 1966 (Menzies and Glynn, 1968). Two years later it was discovered in Venezuela (Glynn, 1970), and in 1978 it was found in Brazil (Pires, 1980). Its first known appearance in the United States was along the coast of Texas (Clark and Robertson, 1982), about the same time it was found in South Carolina (Van Dolah, et al., 1984). P. dianae was then found in 1984 on the Atlantic coast of Florida (Nelson and Demetriades, 1992). Although no records exist of its presence along other portions of the Gulf coast, the Georgia coast or into North Carolina, its establishment in Texas, Florida and South Carolina suggest that it may also occur in those places. Realized and potential adverse impacts: In Florida, P. dianae is one of the most abundant isopods, reaching densities of 53,000 individuals in one square meter (Nelson and Demetriades, 1992). Although the ecology of this species has not been studied in its new home in the South Atlantic Bight (Cape Canaveral to Cape Hatteras), given the high densities noted in Texas and Florida, it may be a significant competitor of other native grazing species for food or for space in the cryptic habitats that itoccupies. Authors:
Cited material: Atta, M.M. 1991. The occurrence of Paradella dianae (Menzies, 1962) (Isopoda, Flabellifera, Sphaeromatidae) in Mediterranean waters of Alexandria. Crustaceana 60: 213-217. Bruce, N.L. 1990. New records of isopod crustaceans (Flabellifera) from Hong Kong. pp. 549-554 In: Morton, B., ed. The marine flora and fauna of Hong Kong and Southern China II. Hong Kong University Press, Hong Kong. Clark, T.S. and Robertson, P.B. 1982. Shallow water marine isopods of Texas. Contribution in Marine Science 25: 45-59. Espinosa-Pérez, M.C. and Hendrickx, M.E. 2001. Checklist of isopods (Crustacea: Peracarida: Isopoda) from the Eastern Tropical Pacific. Belgian Journal of Zoology 131(1): 43-55. Forniz, C. and Maggiore, F. 1985. New records of Sphaeromatidae from the Mediterranean Sea (Crustacea, Isopoda). Oebalia 11: 779-783. Fox, R.S. and Ruppert, E.E. 1985. Shallow-water marine benthic macroinvertebrates of South Carolina. University of South Carolina Press, Columbia, SC.. 329 pp. Furlani, D.M. 1996. A guide to the introduced marine species in Australian waters. Centre for Research on Introduced Marine Species Technical Report No. 5. García-Guerrero, M. and Hendrickx, M.E. 2005. Fecundity and reproductive period of Paradella dianae and Uromunna sp. (Peracarida, Isopoda) associated with prop roots of Rhizophora mangle in a tropical coastal lagoon, SE Gulf of California, Mexico. Crustaceana 78(7): 769-780. Glynn, P.W. 1970. A systematic study of the Sphaeromatidae (Crustacea: Isopoda) of Isla Margarita, Venezuela, with descriptions of three new species. Memoria de la Sociedad de Ciencias Naturales La Salle 85(30): 5-48. Harrison, K. and Holdich, D.M. 1982. Revision of the genera Dynamenella, Ischyromene, Dynamenopsis, and Cymodocella (Crustacea: Isopoda), including a new genus and five new species of eurabranchiate sphaeromatids from Queensland waters. Journal of Crustacean Biology 2: 84-119. Iverson, E.M. 1974. Range extensions for some California marine isopod crustaceans. Bulletin of the Southern California Academy of Sciences 73: 164-169. Javed, W. and Ahmed, R. 1987. On the occurrence of Paradella dianae (Menzies, 1962) a genus and species of Sphaeromatidae (Isopoda, Flabellifera) in the Arabian Sea. Crustaceana 53: 215-220. Kensley, B. and Schotte, M. 1989. Guide to the marine isopod crustaceans of the Caribbean. Smithsonian Institution Press, Washington, D.C. 308 pp. Kensley, B., Schotte, M. 1999. New records of isopods from the Indian River Lagoon, Florida (Crustacea: Peracarida). Proceedings of the Biological Society of Washington 112(4): 695-713. Menzies, F.J. and Frankenberg, D. 1966. Handbook on the common marine isopod Crustacea of Georgia. University of Georgia Press, Athens, Georgia. 93 pp. Menzies, F.J. and Glynn, P. 1968. The common marine isopod Crustacea of Puerto Rico: a handbook for marine biologists. Studies on the Fauna of Curaçao and other Caribbean Islands 27(104): 1-133. Nelson, W.G. and Demetriades, L. 1992. Peracarids associated with sabellariid worm rock (Phragmatopoma lapidosa Kinberg) at Sebastian Inlet, Florida, U.S.A. Journal of Crustacean Biology 12: 647-654. Pires, A.M.S. 1980. New record of Sphaeromatidae (Isopoda) from the Brazilian southern coast: Dynamenella dianae (Menzies, 1962). Crustaceana 39(2): 133-140. Rodriguez, A., Drake, P. and Arias, A.M. 1992. First records of Paracerceis sculpta (Holmes, 1904) and Paradella dianae (Menzies, 1962) (Isopoda, Sphaeromatidae) at the Atlantic coast of Europe. Crustaceana 63: 94-97. Van Dolah, R.F., Knott, D.M. and Calder, D.R. 1984. Ecological effects of rubble weir jetty construction at Murrells Inlet, South Carolina – Vol. I: Colonization and community development on new jetties. Technical Report EL‑84‑4, prepared by Marine Resources Research Institute, Charleston, SC, for Coastal Engineering Research Center, U.S. Army Engineer Waterways Experiment Station, Vicksburg, Mississippi. 138pp. Previous PDF versions of Featured Species (click on title)
|
|||||||